When a pH sensor is placed in a water-based solution, an electro-chemical reaction takes place. A low voltage (mV) signal is generated and conducted through signal wires that connect the sensor to the analyzer/transmitter. Internally, the analyzer “draws a line” based on the input signals. This “line” is the pH curve. The analyzer plots points on the line that correspond to input signal levels. A 7 pH buffer will produce a 0 mV signal, our calibration zero-point. A 4 pH buffer will produce a +180 mV signal, our calibration span point. The analyzer calculates this information, “connecting the dots” with its program. The analyzer also generates usable outputs like a display or a relay activation or a current output. The outputs can be wired to pumps, valves or other equipment.
For the signals to have a real-world meaning, they must be referenced to known standards. In our video, we refer to “calibration”. Calibration is the process of programming the pH analyzer to a known reference (like buffer solution).
When a new sensor is connected to an analyzer, it must be calibrated before use. For analyzers that accept multiple sensor inputs, calibration should be performed for each sensor to ensure accurate, repeatable readings. Regular re-calibration is also necessary. It is important to note that sensor(s), cable(s) and analyzer should be calibrated together as one system for best accuracy.
Analyzers with auto-recognition features enable the appropriate calibration screens to allow calibration for any single-sensor configuration or dual-sensor configuration of the analyzer.
Using auto-calibration instead of manual calibration often avoids common pitfalls in procedure and reduces errors. The analyzer automatically recognizes the buffers and uses temperature-corrected pH values in the calibration. However, due to process conditions, auto-calibration does not work in all cases.
We recommend manual calibration of the pH analyzer using a 2-point method. This means that the sensor will first be rinsed off, dried, placed in a 7 pH (neutral) buffer, programmed, rinsed, dried, placed in a 4 pH (acidic) buffer, programmed, completing the calibration. As mentioned in other notes, pH 4 and pH 7 buffers are the most stable and have the longest shelf life.
Once the pH sensor is placed in a buffer, allow time for the reading to stabilize. The temperature of the sensor is adjusting to the temperature of the buffer. We call this point equilibrium.
Most pH analyzers follow the same methods for calibration. To Manually Calibrate a pH loop on your analyzer, choose 2-point buffer calibration on the calibration menus. Select Manual Buffer entry and follow the step-by-step procedures displayed on the analyzer screen…
Once the analyzer successfully completes the calibration process, it calculates and displays the calibration slope and offset. (The slope is reported as the slope at 25 °C, which is the reference all pH and ORP analyzers use for comparison.)
Q&A
Why start with a 7 pH buffer?
To “zero” and “span” an instrument makes it “real”. We are relating electrical signals to real-world values. A 7 pH buffer produces 0 mV signal from the pH sensor. This is our zero point on the pH curve. A fresh 4 pH buffer will produce a sensor signal output of approximately +180 mV. More Hydrogen ions are acting in a 4 pH solution, so there is a strong signal. Alternately, a fresh 10 pH buffer will produce a sensor signal output of approximately -180 mV. However, there is not as much Hydrogen ion activity here, so the signal will be lower. Also, 10 pH buffers are not very shelf-stable, so it’s best to use them only once. We recommend 7 and 4 buffers.
How does temperature effect pH calibration?
If the temperature fluctuates, the calibration will not be accurate.
pH Slope degrades more in applications with elevated temperatures (greater than 77oF).
What about pH Standardization?
pH standardization can be used on pH sensors that have already had a 2-point calibration. Standardization can help compensate for the effects of pH sensor aging without changing the slope. It is also used to match exact readings with other pH sensors.
Are there any recommendations on the shelf life of pH sensors?
The shelf life for a pH/ORP sensor is one year.
Long-term storage (beyond one year) for any pH sensor is not recommended. Here’s why: the sensor electrolyte solution has a tendency to crystalize. Also, pH glass electrodes may slowly deteriorate in storage.
That being stated, it makes sense to keep a few spares on hand for emergencies (or supplier shortages).
What’s the best way to store pH/ORP sensors?
If you have to store a pH/ORP sensor, make sure to follow these guidelines:
- Store sensors in their original box/shipping containers until needed.
- Do not store sensors at temperatures below 14oF (-10oC).
- Store sensors with their protective cap containing KCL solution, (such as Rosemount p/n 9210342).
If a sensor has been stored for a long time, can we just calibrate it and put it in the process?
Before calibrating, first immerse the sensor in 4 pH buffer solution. This allows the sensor glass to become acclimated for use. Next, calibrate using the 2-point method prior to use.
What about new sensors or those pulled out of a process?
The cap with KCl may dry over time. If the electrolyte solution has crystalized, try rejuvenating the sensor by soaking the sensor in 4 pH buffer overnight. Afterward, perform a 2-point buffer calibration. Check for Slope value. A good, working sensor should have a slope of at least 54 mV/pH.
Do some sensors have longer shelf-life than others?
In the case of Rosemount, all pH/ORP sensor models have the same shelf life.
How does pH slope relate to pH sensor life?
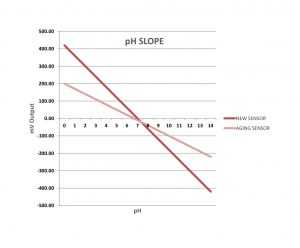
The slope is the indicator of pH sensor life. It is a ratio of sensor response in mV to a corresponding pH level.
As pH glass ages or references become contaminated with the process fluid, the analyzer will receive sensor mV levels that vary from the original calibration curve values. This offset is reflected in the pH slope reading. If you were to graph the curve of the new pH sensor, and the curve of the aging sensor, the slope of each line would be quite different. Eventually, the slope will “flatten out”. At this point, either the junction or sensor should be replaced.
Slope ranges used in pH sensor maintenance:
- Good Slope: 54-59 mV/pH.
- Borderline Slope: 47-50 mV/pH range. Sensor is nearing end-of-life. Change sensor junction, and fill solution. This will extend sensor life. Order a replacement sensor.
- Low Slope: 43-44 mV/pH. It’s time to replace the sensor